Introduction
Background
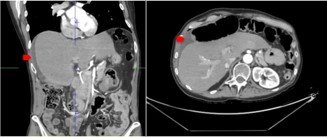
Postoperative pain following cholecystectomy for chronic cholecystitis with gallstones and complicated biliary colic is common. Anatomopathological examination confirmed fibrosis with adenomyosis, without evidence of dysplasia or malignancy. The patient was hospitalized for pain control; however, by postoperative day 4, the patient continued to experience significant pain. A CT scan revealed a small amount of fluid in the perihepatic space, consistent with biliperitoneum. There was no bile duct dilatation or clinical evidence of hemobilia (Quincke triad). A postoperative bilioma was suspected [1]. Exploratory laparoscopy identified bile leakage from the gallbladder bed, originating from an eschar.
Relevant literature context
Several classification systems guide the management of biliary duct injuries. The Strasberg system (modified BismuthCollette) categorizes injuries without vascular involvement, including partial right and left duct injuries. Lau’s classification incorporates associated vascular lesions, and the Stewart–Way system considers anatomy, mechanism, and vascular involvement. These frameworks provide a structured approach to the diagnosis and treatment of biliary duct injuries.
Case presentation
Clinical findings and timeline
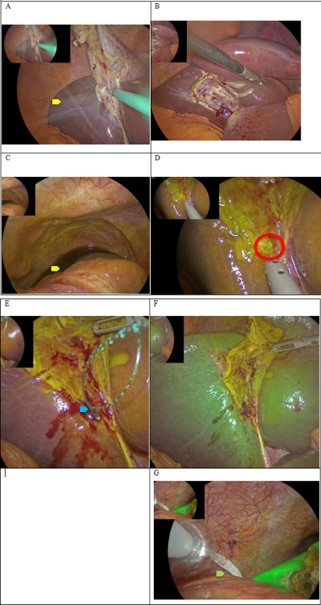
A 60-year-old woman in good health, with no prior surgical history and known asymptomatic gallstones, presented to the emergency room with worsening severe biliary colic and a positive Murphy sign. Laboratory tests and abdominal ultrasound confirmed acute cholecystitis, indicating the need for surgical intervention. On postoperative day 4, despite analgesic treatment, the patient experienced acute right upper quadrant pain and anorexia. A CT scan revealed perihepatic fluid, raising suspicion for a postoperative bilioma. Exploratory laparoscopy demonstrated biliperitoneum. After careful identification of the hepatic anatomy, including Rouviere’s sulcus and the critical view of safety, the source of bile leakage was identified as an aberrant Luschka duct in the gallbladder bed.
Diagnostic assessment
Diagnosis was based on postoperative imaging showing perihepatic fluid collection and clinical presentation of persistent right upper quadrant pain. Intraoperative identification of the aberrant Luschka duct confirmed the source of bile leakage.
Therapeutic intervention
The aberrant Luschka duct was treated with primary suture and clipping during exploratory laparoscopy. A drain was placed for postoperative monitoring.
Follow-up and outcomes
Treatment of the Luschka duct resulted in favorable clinical recovery. No bile output was observed through the right subphrenic drain. Follow-up showed no evidence of late complications, such as stricture, stenosis or cholangitis.
Discussion
Postoperative subphrenic fluid collection following cholecystectomy, as detected on postoperative day 4, may indicate a bilioma, particularly in patients presenting with right upper quadrant peritoneal irritation. Clinically significant bile leaks occur in approximately 0.4–1.2% of cholecystectomies [2]. The diversity of biliary anatomy and patterns of injury necessitate classification systems to guide systematic assessment and management. This underscores the ongoing debate regarding the routine use of intraoperative cholangiography, even among experienced surgeons [3]. Timing of recognition may be early (within 6 weeks) or late (after 6 weeks), and imaging modalities such as CT or MRCP should be performed to guide therapeutic planning.
Currently, there are no universally systematized guidelines. The WSES 2000 guidelines on biliary duct injury provide a seven-question framework for detection and management. The critical view of safety is recommended to minimize bile duct injury. When dissection is unsafe, subtotal cholecystectomy— either reconstituting or fenestrating the gallbladder—may be considered. Evidence supporting conversion to open surgery, intraoperative cholangiography, or indocyanine green imaging is limited. Severe biliary pain and early symptom onset may guide surgical indication, although recommendations remain weak. Once a bile duct injury is identified, drainage should be placed, and referral considered within 48 hours. The Lau classification assists in assessing associated vascular injuries, which informs surgical decision-making. Injuries to the right hepatic artery may be managed by direct repair or observation, while injuries involving the porta hepatis and liver may require repair or hepatectomy.
Bile duct injuries should be stratified as minor or major. Major injuries generally require early definitive repair, such as Roux-en-Y hepaticojejunostomy with drainage and lavage. Minor injuries may be managed with a Kehr T-tube or direct primary suturing, accompanied by drainage. Percutaneous drains may also be used. In cases of postoperative jaundice, ERCP with sphincterotomy and stent placement can alleviate symptoms. Antibiotic prophylaxis should follow guidelines, typically using piperacillin/tazobactam or ceftriaxone with metronidazole for at least five days [4].
Limitations of this case include the absence of MRCP prior to surgery due to the emergency laparoscopic intervention. In other settings, MRCP, ERCP, percutaneous transhepatic cholangiography, or intraoperative cholangiography could help establish the diagnosis and classify injuries according to the Strasberg system (types A–E) [5,6].
Early recognition and accurate classification of bile duct injury are essential. Imaging modalities such as CT, MRCP, and hepatobiliary scintigraphy are important for detecting subtle or delayed injuries. In hemodynamically stable patients, nonoperative management is appropriate for minor liver trauma and small bile leaks, many of which resolve with observation. Major leaks typically require ERCP with biliary stenting. Trauma guidelines support nonoperative care in stable patients while maintaining readiness for urgent surgical intervention if deterioration occurs. Surgical techniques to control liver trauma include the Pringle maneuver with a Rumel tourniquet, tractotomy for deep lesions, omental patching, and balloon tamponade using Foley or Sengstaken catheters. Persistent bleeding after packing and Pringle maneuver may indicate aberrant hepatic artery anatomy or retrohepatic IVC injury. In gallbladder trauma, cholecystectomy is generally required. Complications such as bile peritonitis, biloma, abscess, or stricture demand multidisciplinary management, including drainage, endoscopic intervention, or surgical repair. Long-term risks, including strictures and recurrent cholangitis, are common [7].
Lesions of the hepatic arterial supply, although rare due to extensive liver perfusion, can cause ischemic necrosis. Sudden disruption of intrahepatic blood flow offers less collateral circulation than isolated hepatic artery injury, which may complicate outcomes [8].
Postoperative prophylaxis and early mobilization are critical. Low-Molecular-Weight Heparin (LMWH) reduces the risk of Venous Thromboembolism (VTE), Deep Vein Thrombosis (DVT), and pulmonary embolism (PE), and is preferred over unfractionated heparin. PT/INR is not a reliable predictor of bleeding risk, and direct oral anticoagulants are generally contraindicated. Overall, patients with liver trauma or major hepatobiliary injury are at high risk of thromboembolic complications due to hypercoagulability and immobility. Early enteral feeding and mobilization are safe in stable patients and support recovery without increasing bleeding risk or failure of nonoperative management [9].
Conclusion
Early recognition of bile duct injuries is crucial, as grading these injuries guides therapeutic management by distinguishing minor from major lesions. Knowledge of biliary anatomy, including anatomical variants, and assessment of associated vascular injuries help stratify the severity of the lesion. Imaging is mandatory to localize and characterize the suspected bile duct injury. In cases managed by surgeons without hepatobiliary expertise, prompt transfer to a specialized referral center is recommended. The critical view of safety during cholecystectomy helps minimize both vascular and bile duct injuries. Compared with liver trauma, failure to appropriately manage bile duct injuries can lead to severe complications such as liver necrosis. Other postoperative complications—including bile duct strictures, stenosis, and cholangitis—should be monitored, and early radiologic evaluation is indicated when these are suspected.
References
- Batsuuri B, Lkhagvasuren M. Duct of Luschka: a case report. Ann Hepatobiliary Pancreat Surg. 2023; 27: S373.
- Schnelldorfer T, Sarr MG, Adams DB. What is the duct of Luschka?—a systematic review. J Gastrointest Surg. 2012; 16: 656–662.
- Garcia S, Concepción AM, Wakoff C. Bile leak due to Luschka duct injury after laparoscopic cholecystectomy: a case report. Cureus. 2023; 15: e28427.
- van Dijk AH. Evidence-based medicine in complicated and uncomplicated gallstone disease [thesis]. Amsterdam: University of Amsterdam. 20xx.
- Abdelgawad MS, Eid M, Abokoura S, Elmazaly MA, Aly RA. Iatrogenic bile duct injuries after laparoscopic cholecystectomy: evaluation by MRCP before management. Egypt Liver J. 2023; 13: 2.
- Nasa M, Sharma ZD, Gupta M. Bile duct injury—classification and prevention. J Digest Endosc. 2020; 11: 182–186.
- Puri R. Liver trauma: WSES 2020 guidelines. World J Emerg Surg. 2020; 15: xx–xx. Available from: https://gmka.org/en/articles/ travma-pechinky/
- Coccolini F, Coimbra R, Ordonez C, et al; WSES Expert Panel. Liver injury. World J Emerg Surg. 2020; 15: 24.
- Aggarwal A, Puri K, Liangpunsakul S. Acute liver failure. World J Gastroenterol. 2014; 20: 5737–5747.