Introduction
Parathyroid Carcinoma (PC) is a rare pathology. Although traditionally estimated to have an incidence of 1%, recent studies report significantly lower figures (0.005%), which may be attributed to stricter diagnostic criteria or underdiagnosis. It typically presents between the fourth and sixth decades of life, with no significant sex differences, unlike benign parathyroid tumors, which are more prevalent in elderly women [1-3].
Its pathogenesis remains largely unknown. Most cases are sporadic; however, approximately 25% of patients harbor mutations in the CDC73/HRPT2 gene (chromosome 1), which encodes parafibromin. Familial forms have also been described, including Multiple Endocrine Neoplasia type 1 (MEN1) and Hyperparathyroidism-Jaw Tumor Syndrome (HPT-JT) [2-5].
A high index of clinical suspicion is essential for diagnosis. This requires a thorough biochemical evaluation of calcium-phosphate metabolism and imaging studies for localization, such as cervical ultrasound, 99mTc-sestamibi scintigraphy, and/ or 18F-choline PET-CT. The treatment of choice is en bloc surgical resection of the affected parathyroid gland, with ipsilateral thyroid lobectomy if necessary. Prophylactic lymph node dissection remains a controversial topic [3,4,6,7].
Objective
To present our institutional experience regarding the surgical management and outcomes of patients treated for parathyroid carcinoma.
Materials and methods
We conducted a retrospective, observational, and descriptive study analyzing cases of parathyroid carcinoma operated between 2015 and 2022 in the Endocrine-Metabolic Surgery Unit of our hospital. Histological diagnosis was based on WHO malignancy criteria, including capsular and vascular invasion, atypical mitoses, and tumor necrosis. The follow-up protocol included quarterly biochemical assessments (serum calcium, PTH), semiannual cervical ultrasound, and PET-CT in cases of suspected recurrence. Two patients were referred from other institutions following evidence of tumor recurrence for surgical salvage. Additionally, a narrative review of the current literature was performed.
Clinical cases
Case 1
A 46-year-old female with a history of parathyroid carcinoma diagnosed in 1996, initially treated with left parathyroidectomy and ipsilateral hemithyroidectomy. During follow-up at her local hospital, she underwent three additional surgeries due to local recurrences, identified by persistent hypercalcemia (12.6 mg/ dL), elevated serum PTH (284 pg/mL), and symptoms of fatigue and osteoporosis. Each recurrence was managed surgically, with histopathological confirmation of parathyroid carcinoma. Postoperative biochemical normalization was achieved (calcium 10 mg/dL, PTH 77 pg/mL).
After six years of endocrinology follow-up, new biochemical alterations emerged (calcium 14 mg/dL, PTH 240 pg/mL) along with osteoporosis, prompting referral to our institution. Imaging studies (ultrasound and 99mTc-sestamibi scintigraphy) revealed two retroesophageal nodules, which were surgically excised.
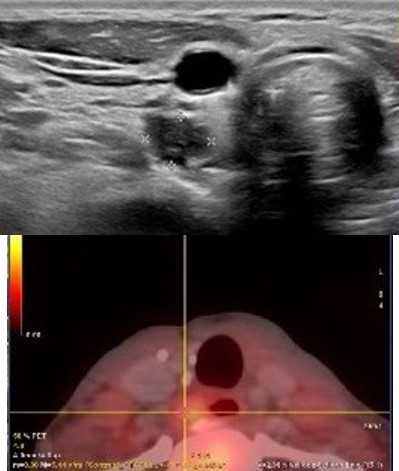
The patient remained disease-free for ten years until new symptoms and biochemical abnormalities reappeared (PTH 229.2 pg/mL, calcium 11.7 mg/dL). Cervical CT and 18F-choline PET-CT identified a hypermetabolic paraesophageal lesion (SUVmax 2.8, 6×7 mm) and three nodules in the upper right thyroid lobe (Figure 1).
Elective surgery was performed, including excision of the affected tissue. Intraoperatively, a paraesophageal nodule was found adherent to the right recurrent laryngeal nerve. Postoperative recovery was uneventful, with normalization of serum calcium levels.
Histopathological analysis confirmed recurrent parathyroid carcinoma with clear resection margins and no tumor infiltration in the paratracheal tissue or excised lymph nodes.
During follow-up, calcium and PTH levels remained within normal range. However, the patient developed dysphonia and liquid dysphagia. Laryngoscopy revealed right recurrent nerve paralysis and reduced mobility of the left vocal cord. She underwent speech therapy, achieving full vocal cord function recovery.
Currently, the patient remains disease-free after ten years of follow-up, with no biochemical or imaging evidence of recurrence.
Case 2
A 56-year-old female was diagnosed with severe hyperparathyroidism in 2016, presenting with asthenia, anorexia, and prolonged vomiting. Laboratory tests revealed marked hypercalcemia (15.7 mg/dL) and elevated PTH (1630 pg/mL). Cervical ultrasound identified a left parathyroid nodule, and left parathyroidectomy was performed. Histopathology reported a parathyroid adenoma. Postoperatively, the patient developed hungry bone syndrome.
During endocrinology follow-up, calcium and PTH levels remained stable until July 2017, when new biochemical alterations were detected (calcium 10.5 mg/dL, PTH 120 pg/mL), suggestive of tumor recurrence. Imaging (ultrasound, 99mTc-sestamibi scintigraphy, and cervical CT) revealed hypermetabolic nodules in the left thyroid lobe. Surgical management included left hemithyroidectomy, remaining ipsilateral parathyroidectomy, and level VI lymph node dissection due to locoregional disease progression. Histopathology confirmed parathyroid carcinoma with extracapsular invasion and involvement of 4 out of 6 lymph nodes. Postoperative complication included left recurrent nerve injury with dysphonia.
The patient was later referred to our center due to a palpable left jugular lymph node suggestive of recurrence. Multidisciplinary evaluation led to left paratracheal and perirecurrent lymph node dissection (level VI). Postoperative recovery was uneventful, with normalized calcium levels.
Due to disease progression, periodic PET-CT follow-up was maintained. Two years later, new hypermetabolic lymph nodes were identified, along with retroesophageal and prevertebral nodules, and biochemical relapse (calcium 13.5 mg/dL, PTH 527 pg/mL).
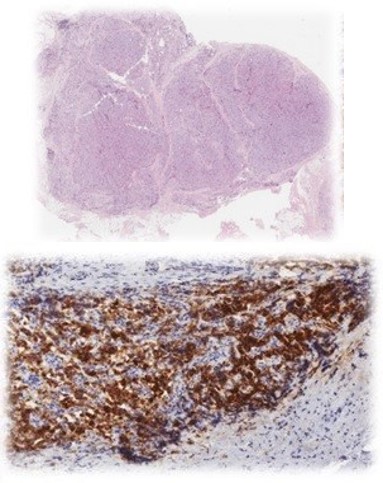
Surgical excision was considered. Fine-needle aspiration (FNA) of the paraesophageal node revealed mature lymphoid cells with low cellularity. Based on clinical and imaging findings, left lymph node dissection was performed. Histopathology confirmed parathyroid carcinoma in infraclavicular and retrocarotid lymph nodes (Figure 2).
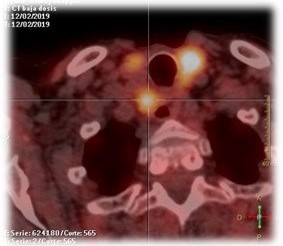
Two months postoperatively, PET-CT revealed persistent hypermetabolic cervical and mediastinal lymphadenopathy and a retroesophageal prevertebral nodule (Figure 3). Due to extensive prior surgeries and persistent disease, radiotherapy was initiated following multidisciplinary consensus.
The patient remains under active surveillance with stable calcium and PTH levels (calcium 10.9 mg/dL, PTH 526.2 pg/mL), receiving cinacalcet and zoledronic acid. PET-CT shows four stable hypermetabolic lymph nodes in retroesophageal/prevertebral, retroclavicular, prevascular, and lower right paratracheal locations.
Case 3
An 87-year-old female presented with asthenia, anorexia, vomiting, nocturia, and urinary frequency. Laboratory tests revealed severe hypercalcemia (24 mg/dL) and markedly elevated PTH (3,157 pg/mL). Densitometry showed diffuse osteopenia, and skull X-rays revealed multiple osteolytic lesions suggestive of myeloma.
Given the extreme biochemical profile and osteolytic findings, hematologic malignancies were initially considered but ruled out by imaging.
Cervical ultrasound, 18F-choline PET-CT, and 99mTc-sestamibi scintigraphy identified a hyperfunctioning parathyroid lesion (6×4 cm) in the left thyroid lobe, extending into the retroesophageal space, mediastinum, and thoracic vertebra T3. Suspecting parathyroid carcinoma, en bloc resection was performed, including four anterior and posterior mediastinal lymph nodes. Sternotomy was required for complete excision.
Histopathology confirmed parathyroid carcinoma with capsular invasion and vascular embolization, without lymph node involvement.
Postoperative recovery was favorable, and the patient was discharged on postoperative day two. During follow-up, no biochemical evidence of recurrence was observed (calcium 8.7 mg/ dL). The patient died several years later due to cardiac causes.
Table 1: Biochemical parameters at diagnosis and recurrence.
| |
PTH (pg/mL) |
Calcio (mg/dL) |
Fósforo (mg/dL) |
| Case 1 |
Diagnosis
Recurrence |
128
229,20 |
11,46
11,70 |
2,14
2 |
| Case 2 |
Diagnosis
Recurrence |
319
527 |
11,7
13,5 |
2,1
1,3 |
| Case 3 |
Diagnosis
Recurrence |
3.157
- |
24
- |
5,30
- |
Discussion
Parathyroid Carcinoma (PC) is a rare entity, accounting for less than 1% of primary hyperparathyroidism cases [4,8]. Peak incidence occurs between 40 and 60 years of age, with no sex predilection. In our series, all three patients were female, two of whom fell outside the typical age range [1,2].
Its pathophysiology remains unclear. CDC73/HRPT2 mutations are detected in 25% of sporadic cases. Genetic testing is currently recommended only for first-degree relatives. Although no direct risk factors have been established, most patients with PC have a family history or prior neck irradiation. None of our patients underwent genetic testing due to the absence of familial or clinical risk criterio [1-5,8].
Most tumors are hormonally active, with biochemical and clinical manifestations such as fatigue, bone pain, and nephrolithiasis. A high index of suspicion for PC is warranted when serum calcium exceeds 14 mg/dL and PTH levels are 5-10 times the Upper Limit of Normal (ULN), in contrast to primary hyperparathyroidism, where calcium typically ranges from 10-12 mg/dL and PTH is up to 2× ULN [4,9]. All patients in our series presented with fatigue, bone pain, nephrolithiasis, and corresponding biochemical and densitometric abnormalities (Table 1).
Its indolent course often leads to lymphatic (15-30%) and distant metastases (33%) at diagnosis, commonly involving lungs, liver, and bone. Only one patient in our series had nodal involvement at initial diagnosis.
Preoperative localization should include at least two concordant imaging modalities, with ultrasound and 99mTc-sestamibi scintigraphy as first-line tools. In cases of suspected distant metastases, CT or MRI may be useful. Fine-Needle Aspiration (FNA) is contraindicated due to the risk of tumor seeding and limited diagnostic utility. 18F-FDG PET-CT is considered secondline when conventional imaging fails, though it may be used as first-line in selected cases [1,2,10,11].
In our series, FNA and PET-CT were selectively applied based on clinical criteria and multidisciplinary consensus. FNA was performed in one case to exclude alternative etiologies, despite its diagnostic limitations.
PC remains a diagnostic challenge. Clinical suspicion should be raised by suggestive findings such as calcium >14 mg/dL, cervical lesions >3 cm, renal (nephrolithiasis, nephrocalcinosis) and skeletal abnormalities (osteoporosis, vertebral fractures). Definitive diagnosis relies on histopathological evaluation (Figure 2).
The surgical gold standard is en bloc parathyroidectomy with ipsilateral hemithyroidectomy and excision of invaded tissues and lymph nodes. Prophylactic lymphadenectomy has not demonstrated survival benefit and is reserved for cases with evident involvement due to its high morbidity [1-3,6].
Common postoperative complications include recurrent laryngeal nerve injury and metabolic disturbances such as hungry bone síndrome [1,8]. Both patients referred from other centers developed these complications after prior surgeries, requiring speech and voice therapy.
The SEER database reports lymph node metastases in 25.2% and distant metastases in 2.2% of cases. In our series, distant metastases were absent, though two patients experienced locoregional recurrence (66%), prompting referral for surgical salvage.
Recurrence rates range from 50-60%, typically occurring between 2.5 and 5 years postoperatively, underscoring the need for prolonged and rigorous follow-up [1,2,4]. Some centers utilize intraoperative infrared parathyroid identification and/or Indocyanine Green (ICG) fluorescence to aid in detecting recurrent or persistent disease [3,7].
ICG angiography is primarily used intraoperatively to identify parathyroid glands in recurrent disease. However, it does not replace Postoperative Surveillance strategies (PET-CT, CT) or contribute to molecular diagnosis [12-14].
Near-infrared autofluorescence imaging (NIRAF/PTeye™) is another modality that enables rapid, non-invasive identification of parathyroid tissue without contrast agents. While highly sensitive and specific, it does not assess gland viability or perfusión [12,14,15].
PTeye™ is considered superior for gland localization, whereas ICG angiography is preferred for perfusion assessment. Both techniques may be used complementarily during surgery [12,14,15].
Although these technologies have shown utility in thyroid surgery, their benefit in PC remains unproven. They were not employed in our series due to lack of direct recommendations in the literature.
Despite PC being considered radioand chemoresistant, one patient in our series received radiotherapy due to persistent disease after multiple surgeries. No effective chemotherapy regimens are currently described, and neither radiotherapy nor chemotherapy has demonstrated survival Benefit [7,9].
In unresectable or metastatic cases, the primary goal is symptomatic control using pharmacologic agents (e.g., mitramycin, calcimimetics) and/or ablative techniques (e.g., radiofrequency, embolization) [1,8].
Five-year overall and disease-specific survival in our series was 100%, exceeding the 85% reported in the literature, likely due to the small sample size. Prognostic factors include presence of metastases, incomplete resection, and parafibromin gene silencing [2,6]. Elevated serum calcium was the most consistent predictor of recurrence across cases.
Conclusion
Parathyroid carcinoma is a rare entity requiring high clinical suspicion and early diagnosis. Thorough preoperative evaluation and precise surgical management are essential. We advocate for referral of suspected PC cases to specialized endocrine surgery centers for multidisciplinary evaluation and individualized treatment.
References
- Wei CH, Harari A. Parathyroid carcinoma: update and guidelines for management. Curr Treat Options Oncol. 2012; 13: 11–23.
- Cetani F, Pardi E, Marcocci C. Parathyroid carcinoma. In: Brandi ML, editor. Frontiers of Hormone Research. Basel: Karger; 2019. 63–76.
- Betea D, Potorac I, Beckers A. Parathyroid carcinoma: challenges in diagnosis and treatment. Ann Endocrinol. 2015; 76: 169–77.
- Barberán M, Campusano C, Salman P, Trejo P, Silva-Figueroa A, Rivera S, et al. Puesta al día: carcinoma paratiroideo. Rev Med Chil. 2021; 149: 399–408.
- Christakis I, Busaidy NL, Cote GJ, Williams MD, Hyde SM, Silva Figueroa AM, et al. Parathyroid carcinoma and atypical parathyroid neoplasms in MEN1 patients: a clinico-pathologic challenge. Int J Surg. 2016; 31: 10–6.
- Asare EA, Sturgeon C, Winchester DJ, Liu L, Palis B, Perrier ND, et al. Parathyroid carcinoma: an update on treatment outcomes and prognostic factors from the National Cancer Data Base. Ann Surg Oncol. 2015; 22: 3990–5.
- Viswanath A, Drakou EE, Lajeunesse-Trempe F, Grossman AB, Dimitriadis GK. Parathyroid carcinoma: new insights. Best Pract Res Clin Endocrinol Metab. 2025; 39: 101966.
- Rodrigo JP, Hernandez-Prera JC, Randolph GW, Zafereo ME, Hartl DM, Silver CE, et al. Parathyroid cancer: an update. Cancer Treat Rev. 2020; 86: 102012.
- Sillero Sánchez A, Atienza Iglesias MA. Manejo diagnóstico-terapéutico del carcinoma de paratiroides. An Med Interna. 2002; 19: 48–52.
- Uljanovs R, Sinkarevs S, Strumfs B, Vidusa L, Merkurjeva K, Strumfa I. Immunohistochemical profile of parathyroid tumours: a comprehensive review. Int J Mol Sci. 2022; 23: 6981.
- Boudousq V, Guignard N, Gilly O, Chambert B, Mamou A, Moranne O, et al. Diagnostic performance of cervical ultrasound, 99mTc-sestamibi scintigraphy, and contrast-enhanced 18F-fluorocholine PET in primary hyperparathyroidism. J Nucl Med. 2022; 63: 1081–6.
- Dip F, Alesina PF, Anuwong A, Arora E, Berber E, Bonnin-Pascual J, et al. Use of fluorescence imaging and indocyanine green during thyroid and parathyroid surgery: results of an intercontinental, multidisciplinary Delphi survey. Surgery. 2022; 172: S6–13.
- Spartalis E, Ntokos G, Georgiou K, Zografos G, Tsourouflis G, Dimitroulis D, et al. Intraoperative indocyanine green angiography for the identification of the parathyroid glands: current evidence and future perspectives. In Vivo. 2020; 34: 23–32.
- St Amour TC, Demarchi MS, Thomas G, Triponez F, Kiernan CM, Solórzano CC. Educational review: intraoperative parathyroid fluorescence detection technology in thyroid and parathyroid surgery. Ann Surg Oncol. 2023; 30: 973–93.
- Silver Karcioglu AL, Triponez F, Solórzano CC, Iwata AJ, Abdelhamid Ahmed AH, Almquist M, et al. Emerging imaging technologies for parathyroid gland identification and vascular assessment in thyroid surgery: a review from the American Head and Neck Society Endocrine Surgery Section. JAMA Otolaryngol Head Neck Surg. 2023; 149: 253–60.