Introduction
Persistent Müllerian Duct Syndrome (PMDS) is a rare genetic condition defined by the presence of Müllerian duct-derived structures (fallopian tubes, uterus, and upper vagina) in an otherwise normal phenotypical and genotypical (46, XY) male. Accurate incidence remains largely unknown. There are less than 300 cases described in the medical literature, with various management options for the condition described [1-3]. It could be incidentally found during orchidopexy, laparotomy, or routine inguinal hernia repair in patients presenting with cryptorchidism (undescended testis). Usually, one testis is typically positioned, and the other is undescended. However, it is conceivable to be bilateral as well [4,5] as in this case. The persistence of Müllerian duct derivatives occur either due to deficiency of Anti-Müllerian Hormone (AMH) or mutations in the AMH type II receptor (AMHR-II). During development of a male fetus, these two proteins work together to induce breakdown (regression) of the Müllerian ducts. The current study documents the case of a 28-year-old male patient who was diagnosed with PMDS per-operatively and presents a brief review regarding the relevant literature on the diagnosis and treatment of PMDS.
Case presentation
A 28-year-old male presented with right-sided painful inguinoscrotal swelling. He had history of swelling since childhood. Now from the last 2 years, the size of the swelling was increasing gradually with an aching sensation in the scrotum.
On examination, he was a normal virilized male. He had a 10 x 8 cm swelling in the inguinoscrotal area which was non tender, reducible with a positive cough impulse and ring occlusion test. Testes were palpable in the scrotum. Secondary sexual characteristics were fully developed and the penis was normal but displaced towards left because of the swelling. The scrotum was also well developed. He was clinically diagnosed to be right indirect inguinal hernia.
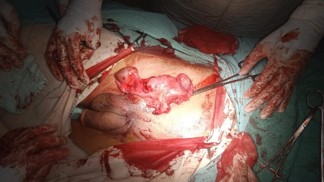
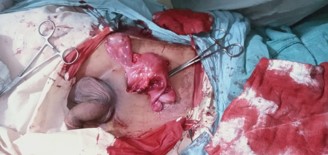
While performing Lichtenstein mesh hernioplasty, hernial sac contained omentum which further revealed uterus with fallopian tubes, upper vagina, and an undescended left tests. The right testes was present in scrotum which was pre operatively conceived to be bilateral (Figures 1 & 2).
Keeping in view the risk of Müllerian and testicular malignancies, Müllerian remnants were excised and left testis was placed in left hemiscrotum by making a rent in median raphe from the right side as the cord structures and blood supply were coming from the right side only. Müllerian remnants were sent for histopathology and hernia was repaired with polypropylene mesh.
The histopathology sections revealed atrophic endometrial glands. Unremarkable myometrium and fallopian tubes with no evidence of malignancy. Postoperatively, the patient underwent hormonal analysis and the results are as follows:
Serum testosterone: 11.3 ng/ml (normal 6.40-31.80) Serum FSH: 9.0 mIU/L
Serum LH: 8.4 IU/L (normal: 1.28.6 IU/L)
The patient is currently in follow up with abdominal ultrasound and scrotal color Doppler planned at 3 and 6 months after surgery and every 6 months thereafter.
The case report is done in accordance with the SCARE criteria.
Discussion
PMDS was first described in a male with an inguinal hernia in 1939 by Nilson, presenting as hernia uteri inguinal [6]. PMDS is a rare form of internal male pseudohermaphroditism caused by a deficiency of MIF [7,8].
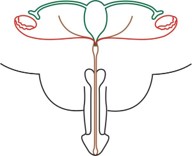
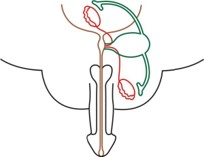
In a human fetus, the Müllerian and Wolffian ducts are both present at 7 weeks of gestation. In a male fetus, the testis differentiates by the end of the 7th gestational week. Normal sex differentiation is controlled by testosterone, dihydrotestosterone, and MIF. Sertoli cells secrete MIF, which leads to regression of the Müllerian ducts. Testosterone has a direct effect on the Wolffian ducts, and promotes their differentiation into the epididymis, vas deferens, and seminal vesicles [8]. Dihydrotestosterone induces male differentiation of external genitalia. PMDS patients have both Wolffian and Müllerian duct structures due to a deficiency of MIF. PMDS usually presents incidentally in a male presenting with cryptorchid testes or inguinal hernia in childhood which is consistent with our case or sometimes in adulthood or testicular tumor or abdominal mass in adulthood [3,4,9]. PMDS patients are rarely fertile and most of them usually suffer from infertility and inguinal hernia. There are various causes of infertility such as testicular hypoplasia or ejaculatory duct obstruction due to compression by MD structures PMDS is classified on the basis of location of testes and Müllerian structures. There are three clinical anatomical variants described in the literature (Figure 3) [10].
In FT PMDS, both testes are intra-abdominal, in the ovarian position, with the vasa and associated vasculature closely applied to the fallopian tube-like structures (Figure 3a). In children, fertility preservation and hormonal function should be a prime objective. Gonadal malignancy risk should be reduced by early orchidopexy, as for any child with intra-abdominal testes [10].
PMDS-Hernia Uteri Inguinale (HUI) is the most common variant encountered for 80-90% of the cases and usually presents with an ipsilateral hernia with ipsilateral descended testis and Müllerian structures within the hernia. The contralateral testis is usually undescended or intraabdominal (Figure 3b) [10]. Our described case had PMDS-HUI.
PMDS-transverse Testicular Ectopia (TTE) is a rare type of PMDS (Figure 3c) [10]. Both testes and the Müllerian structures are herniated into one hemiscrotum.
The diagnosis of PMDS is often controversial, because there are no specific clinical symptoms, it is discovered incidentally either during hernia repair or surgery for undescended testes. The risk of malignant transformation increases after puberty; therefore, early diagnosis should be recommended.
Treatment of the remnants of MD remains debatable. Previous studies have demonstrated that patients with PMDS require removal of MD remnants due to malignant tumors identified in Müllerian remnants and the MD being connected with the seminal vesicle causing urinary tract infections, periodic hematuria, stones and urination disorders [11,12]. However, other studies have suggested retaining Müllerian remnants to prevent damage to the vas deferens and disruption of collateral blood supply to the testes [11,12].
A long-term management strategy includes assessment of testicular function and monitoring of malignant transformation at an earlier stage as well as follow up with Doppler studies and hormonal profile analysis for the patient in which excision of remnants has been done as the diagnosis is often made incidentally during surgery for an inguinal hernia or during exploration for cryptorchidism. Transverse testicular ectopia should be suspected preoperatively in patients who have unilateral inguinal hernia associated with a contralateral nonpalpable testis. In suspected cases, ultrasonography, computerized tomography, magnetic resonance imaging, and laparoscopy may be helpful in diagnosis [13]. Before puberty in patients with bilateral cryptorchidism, serum AMH levels also help in diagnosis.
Conclusion
In conclusion, the incidence and prevalence of PMDS are poorly estimated. Müllerian structures should be removed whenever possible to avoid the risk of malignancy but the need for follow up is longer. Early diagnosis of PMDS makes excision of Müllerian structures and primary orchidopexy possible. Long-term follow-up is also required for patients with intact Müllerian structures, and MRI may be a better imaging method than US for that purpose. Further diagnostic modalities are required in patients who presents with inguinal hernia with contralateral cryptorchidism or those children who have congenital bilateral cryptorchidism for the early detection of PMDS and prevention of possible complications like malignancies.
Patient consent: Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
References
- Farikullah J, Ehtisham S, Nappo S, Patel L, Hennayake S. Persistent Müllerian duct syndrome: lessons learned from managing a series of eight patients over a 10-year period and review of literature regarding malignant risk from the Müllerian remnants. BJU Int. 2012; 110: E1084–E1089.
- Shalaby MM, Kurkar A, Zarzour MA, Faddan AA, Khalil M, Abdelhafez MF. The management of the persistent Müllerian duct syndrome. Arab J Urol. 2014; 12: 239–244.
- Picard JY, Cate RL, Racine C, Josso N. The persistent Müllerian duct syndrome: an update based upon a personal experience of 157 cases. Sex Dev. 2017; 11: 109–125.
- Saleem M, Ather U, Mirza B, et al. Persistent Müllerian duct syndrome: a 24-year experience. J Pediatr Surg. 2016; 51: 1721–4.
- Renu D, Rao G, Ranganath K, et al. Persistent Müllerian duct syndrome. Indian J Radiol Imaging. 2010; 20: 72.
- Nilson O. Hernia uteri inguinalis beim Manne. Acta Chir Scand. 1939; 83: 231.
- Gutte AA, Pendharkar PS, Sorte SZ. Transverse testicular ectopia associated with persistent Müllerian duct syndrome – the role of imaging. Br J Radiol. 2008; 81: E176–8.
- Dekker HM, de Jong IJ, Sanders J, Wolf RF. Persistent Müllerian duct syndrome. Radiographics. 2003; 23: 309–13.
- Al-Asmar A, Abu-Qamar A, Al-Saidah N, Al-Kaabneh A, Al-Qaralleh A, Al-Hiari A. Persistant Müllerian duct syndrome with intra-abdominal seminoma. Urol Case Rep. 2020; 33.
- Persistent Müllerian Duct Syndrome: Understanding the Challenges PMC (nih.gov).
- Patil V, Muktinaini S, Patil R, Verma A. Persistent Müllerian duct syndrome: a case report. Indian J Surg. 2013; 75: S460–S462.
- Manjunath BG, Shenoy VG, Raj P. Persistent Müllerian duct syndrome: how to deal with the Müllerian duct remnants – a review. Indian J Surg. 2010; 72: 16–19.
- David AD. Sexual differentiation: normal and abnormal. In: Wein AJ, Kavoussi LR, Novick AC, Partin AW, Peters CA, editors. Campbell-Walsh urology. 9th ed. Philadelphia: Saunders. 2007: 3826–7.