Introduction
This report presents a successful bilateral Deep Inferior Epigastric Perforator (DIEP) flap breast reconstruction in a patient with systemic mastocytosis—an uncommon and potentially high-risk comorbidity in reconstructive surgery. Through detailed preoperative planning and multidisciplinary coordination, we achieved a favourable outcome despite a condition that may threaten free flap survival.
Case report
A 40-year-old woman with extensive high-grade triple-positive right breast Ductal Carcinoma In Situ (DCIS) presented requesting bilateral mastectomy, with immediate bilateral Deep Inferior Epigastric Perforator (DIEP) flap reconstruction.
Her past medical history is significant for clinically diagnosed adult-onset Indolent Systemic Mastocytosis (ISM) with predominant cutaneous manifestation over the torso and proximal limbs. Symptoms consist of intermittent urticarial rash in response to heat, anxiety, exercise, and alcohol consumption, but no history of anaphylaxis. Past medical history includes Crohn’s disease and iron deficiency anaemia. Surgical history included two caesarean sections under spinal anaesthetic, and one previous general anaesthetic as a child. Current medications include fexofenadine 180mg bd, magnesium and iron supplementation.
Baseline serum tryptase was elevated at 56 mcg/L. Following multi-disciplinary discussions between the surgeon, immunologist, and anaesthetist, it was deemed safe to proceed with autologous free flap breast reconstruction with DIEP using perioperative risk mitigation strategies. In the event of intraoperative mast cell activation, this would be abandoned in favour of breast tissue expander-implant reconstruction pathway.
The patient was prescribed fexofenadine regularly for a week leading up to the day of surgery. On the morning of surgery, intravenous midazolam was administered in the pre-anaesthetic bay, followed by intra-arterial cannulation for haemodynamic monitoring using the EV1000 monitoring system (Edwards Lifesciences, Irvine, CA, USA). General anaesthesia was then induced with 100 mcg of fentanyl, propofol using target-controlled infusion (TCI) (Schneider model, effect site) and 50 mg of rocuronium. The trachea was intubated. Anaesthesia was maintained with propofol (target concentration 2.5-3.5 mcg.mL1). Her baseline temperature was 36.3°C, and it was monitored closely using an oropharyngeal temperature probe. A warming blanket was placed over the lower half of her body. She was given 8 mg of dexamethasone and 1 mg of tranexamic acid prior to starting.
Bilateral skin sparing mastectomies were performed and simultaneously, DIEP flaps were elevated, guided by pre-operative CTA planning and bedside Doppler. Both flaps were raised on a single perforator. Following completion of the mastectomies, internal mammary vessels were prepared after removal of the 3rd costal cartilage. Arterial anastomosis was performed in the anterograde internal mammary arteries with 8.0 nylon while Synovis venous couplers (2.0 mm right and 2.5 mm left) were used for anterograde internal mammary venous anastomosis. The breast reconstructions were closed over drains. The abdominal flap was quilted and resultant defect closed without drains. The standard free flap monitoring protocol was followed without incident. The flap raise and microvascular anastomoses were more challenging than normal due to persistent ooze.
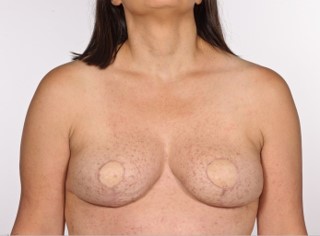
This increased bleeding intra-operatively correlated with hypofibrinogenaemia (1.7 g/L). There was a slow decline in haemoglobin from 110 g/L to 76 g/L. Her temperature had risen to 37.5°C. Serum tryptase was stable at 50 mcg/L. Five units of cryoprecipitate and one unit of packed red cells were given to good effect. The warming blanket was turned off. The thromboelastogram (TEG) was normal at the end of the operation, although the temperature remained elevated at 37.9°C. Intraoperative analgesia comprised of ketamine, lignocaine, and clonidine. The total operating time was 11 hours, after which she was extubated and transferred to the ward. Her hospital stay was uneventful, and she was discharged after five days. The recovery period following discharge was uneventful. The patient was followed up in clinic at 12 weeks (Figure 1).
Discussion
Mastocytosis is a rare chronic myeloproliferative disorder of the mast cells, with a prevalence of 1 in 150,000 [1]. The propensity for mast cell activation and degranulation is elevated in patients with mastocytosis, featuring a non-immunoglobulin E (IgE)-mediated reaction in response to non-specific triggers such as psychological, mechanical, pharmacological, and temperature-related factors. The end result is the release of mediators such as histamine, proteases and cytokines, which result in profound vasodilation and increase in capillary permeability, causing cardiovascular compromise [1-4]. This could theoretically have drastic implications on the survival of a free flap, in the case of our patient, bilateral DIEP flaps.
The focus of perioperative management is the prevention of mast cell activation. In our case, there was an elevated basal tryptase, which is an indicator of the baseline mast cell activation. Although there have been recommendations to provide anti-histamine therapy and steroid cover perioperatively, there is no robust evidence regarding their efficacy in prevention of mast cell activation [3-5]. Nonetheless, histamine-releasing drugs should be avoided, and there have been case reports of fatality with their use as part of anaesthesia in these patients [6,7]. Psychological triggers such as anxiety need to be managed appropriately, and care must be taken to avoid pressures injuries. Temperature control is important, and we postulate that the rise in temperature may have caused a low-grade mast cell activation in our case, resulting in hypofibrinogenaemia and consequent venous ooze. This was managed effectively with cessation of patient warming and administration of cryoprecipitate.
To our knowledge there has not been a free-flap reconstruction described in the presence of systemic mastocytosis. The challenge presented here is the propensity for mediator release to affect flap perfusion, resulting in ischaemia, venous engorgement, tissue oedema and clot formation. The risk for this would be heightened in our case due to the presence of cutaneous mastocytosis in the breast. Therefore, careful multi-disciplinary discussions, appropriate planning, and shared decision-making about the risks involved are vital. In this case, the patient was counselled, and the potential risks outlined; a back-up option of tissue expanders was planned. Intraoperatively, as the serum tryptase remained stable, haemodynamics were unchanged, and the ooze settled after cryoprecipitate therapy, we proceeded with DIEP reconstruction.
Post-operatively, there were no issues with flap perfusion, however, vigilance is key. Awareness of the triggers for mast cell activation is important throughout the perioperative period. The regular use of post operative warming was avoided in this specific case. Appropriate education of the nursing staff is necessary given the rarity of this condition.
Conclusion
Systemic mastocytosis creates a myriad of challenges to both the anaesthetist and the surgeon, especially during freeflap reconstruction surgery though is not a contraindication to this type of surgery. A good understanding of this disorder, its triggers, and prevention strategies, are therefore important in order to ensure free-flap survival.
References
- Dewachter P, Castells MC, Hepner DL, Mouton-Faivre C. Perioperative management of patients with mastocytosis. nesthesiology. 2014; 120: 753-9.
- Burlacu CL, ffrench-O’Corroll R, Morrison C. Considerations in providing safe anaesthesia in a patient with mastocytosis. BJA: British Journal of Anaesthesia. 2014: 113.
- Bocca-Tjeertes IFA, Ven AAJM, Koppelman GH, Sprikkelman AB, Oude Elberink HJNG. Medical algorithm: Peri-operative management of mastocytosis patients. Allergy. 2021; 76: 3233–5.
- Chaar CIO, Bell RL, Duffy TP, Duffy AJ. Guidelines for safe surgery in patients with systemic mastocytosis. The American surgeon. 2009; 75: 74–80.
- Hermans MAW, Arends NJT, Gerth van Wijk R, van Hagen PM, Kluin-Nelemans HC, Oude Elberink HNG, et al. Management around invasive procedures in mastocytosis: An update. Ann Allergy Asthma Immunol. 2017; 119: 304-9.
- Vaughan ST, Jones GN. Systemic mastocytosis presenting as profound cardiovascular collapse during anaesthesia. Anaesthesia. 1998; 53: 804-7.
- Desborough JP, Taylor I, Hattersley A, Garden A, Wolff A, Bloom SR, Morgan M. Massive histamine release in a patient with systemic mastocytosis. Br J Anaesth. 1990; 65: 833-6.
- Lau, S., Sprung, J., Volcheck, G.W. et al. Perioperative management of mastocytosis. J Anesth. 2023; 37: 741–748.