Introduction
Gastrointestinal Stromal Tumor (GIST) is a rare mesenchymal tumor of the gastrointestinal tract, with characteristics varying from benign to malignant. This kind of tumor usually originates from the muscular layer cells and mesenchymal stem cells of the gastrointestinal tract wall and can occur either inside or outside the gastrointestinal tract, previously often misdiagnosed as smooth muscle tumors or neurogenetic tumors, etc. GIST mainly occur in the stomach (55%), small intestine (31%), colorectum (6%), esophagus (<1%), or extra-gastrointestinal organs (<5%). There are three histopathological subtypes of GIST: spindle cell type (77%), epithelioid cell type (8%), and mixed type (15%) [1]. Extra-gastrointestinal stromal tumors (EGIST) primarily originating from the bladder are extremely rare. Nine cases of bladder EGIST patients have been reported previously, including three men and three women, with ages ranging from 15 to 78 years old [2-10]. Here we report a case of EGIST with a huge tumor volume and full-thickness bladder infiltration, without lymphatic and distant metastases.
Case presentation
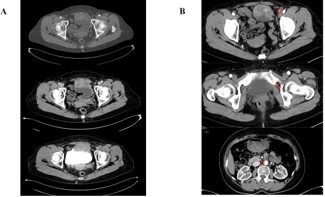
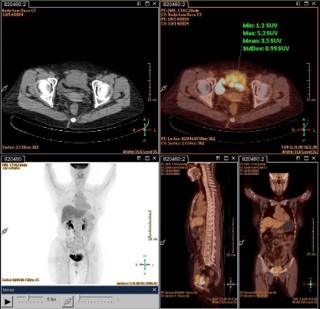
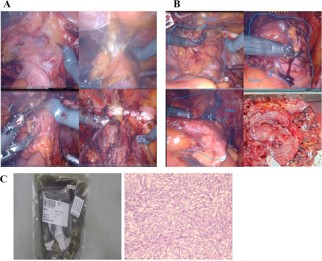
A 64-year-old woman presented with recurrent lower abdominal distension and discomfort for one week. After undergoing a pelvic CT scan, the results showed a lesion in the pelvis, a mass in front of the bladder, and it was considered to be a tumor with a high probability of malignancy (Figure 1A). The boundary between the lesion and the abdominal muscle on the front side was unclear, and there was a possibility of a vascular tumor thrombus on the left side (Figure 1B). There was a possibility of metastasis to multiple lymph nodes on the left side of the iliac external, obturator, and lateral branches of the aorta. In September 2024, due to the unclear nature of the lesion, we first performed a transurethral resection of the bladder lesion under general anesthesia to establish a definitive diagnosis and alleviate the patient’s sensation of abdominal distension. The postoperative pathological results indicated that it was a malignant spindle cell tumor of the bladder. The results of postoperative immunohistochemistry showed: UP3 (-), Myogenin (-), MyoD1 (-), CK (-), VIM (+), SMA (partially +), KI-67 (approximately 60% +), GATA3 (-), DESMIN (-). Two weeks later, the patient experienced urethral bleeding after urination without any obvious predisposing cause, accompanied by mild low back pain. After a comprehensive PET-CT examination, the results indicated a space-occupying lesion on the anterior wall of the bladder with increased metabolism, suggesting a tumor. It has not involved the gastrointestinal tract and there is no evidence of distant metastasis (Figure 2). In October 2024, a robotic-assisted laparoscopic radical cystectomy + ileal conduit operation was performed under general anesthesia. Intraoperatively, we identified this neoplasm originating from the anterior wall of the bladder, which was adherent to the abdominal wall muscles (Figure 3A). The neoplasm was large in size, grotesquely shaped, and soft in texture, resembling a cord-like structure of the intestine. It had invaded the extra-bladder fat, and tumor thrombi were found within the lumens surrounding the bladder. Upon postoperative examination of the specimen, the bladder measured approximately 8.0 x 9.0 x 2.5 cm in size. A cord-like mass measuring about 7.0 x 5.0 x 3.0 cm was visible on the anterior wall of the bladder, with a gray-brown cut surface that was soft in texture (Figure 3B). Upon repeat pathological analysis, the results unequivocally indicated that this is a malignant spindle cell tumor (Figure 3C). Immunohistochemistry indicated: CKH (-), CKL (-), EMA (-), CAM5.2 (-), VIM (+), SMA (+), CK5/6 (-), P40 (-), GATA-3 (-), Desmin (-), ALK (-), UP-3 (-), KI-67 20% (+), S-100 (-), CD34 (+), CD68 (+), ER (-), PR (-), SS18 (-), Calponin (-), MC (-), CR (-), WT-1 (-), CD10 (-), MDM-2 (-), CDK4 (-), CD117 (+), DOG-1 weakly (+), Caldesmon weakly (+), STAT6 (-) (Figure 2B). Combined with the results of immunohistochemistry, the specimen was consistent with extra-gastrointestinal stromal tumor (the maximum diameter of the tumor is approximately 7 cm, the mitotic figure is about 3 per 50 high-power fields, high risk grade).
Discussion
GISTs originate from the Interstitial Cells of Cajal (ICC) and mesenchymal stem cells homologous to Cajal cells within the muscularis layer of the Gastrointestinal (GI) tract wall. They can also occur in extra-gastrointestinal locations within the abdominal cavity, previously often misdiagnosed as smooth muscle tumors or neurogenetic tumors. These tumors are composed of varying proportions of spindle cells and epithelioid cells [1]. The main differential diagnoses for spindle cell-like EGISTs in the bladder are smooth muscle tumors (leiomyoma and leiomyosarcoma), while for epithelioid EGISTs, the differential diagnoses include poorly differentiated carcinomas, neuroendocrine tumors, and epithelioid leiomyosarcomas [1]. In this instance, we identified a GIST localized to the anterior wall of the bladder, with a grotesque, cord-like intestinal appearance and a soft texture, and the final pathological results confirmed it to be a spindle cell-type GIST.
Genetic mutation testing is of significant importance for the diagnosis and treatment of GISTs. Approximately 80-85% of GISTs harbor c-kit gene mutations, and another 10% have PDGFRA gene mutations [11]. Additionally, there are wild-type GISTs, including SDH-deficient, NF1-associated, BRAF-mutated, and KRAS-mutated types. Based on similarities in immunophenotype, ultrastructure, and cellular signaling, GISTs are believed to originate from KIT+ ICCs through somatic KIT mutations, which is considered an early step in the pathogenesis of GISTs [12]. PDGFRA-mutated stromal tumors may originate from telocytes [13]. Animal experiments confirm that BRAF-mutated stromal tumors are more likely to be of smooth muscle cell origin [14].
In this case, CD117 (+), CD34 (+), and weak DOG-1 (+) are crucial evidence for the diagnosis of EGIST [15], also indicating that this EGIST is of the KIT-mutated type. The proto-oncogene c-kit is located on human chromosome 4q11-12, and its product is CD117. However, in GISTs carrying c-kit gene mutations, the positive rate for DOG-1 is 92%, and for CD117, it is 81%, making DOG-1 a relatively more sensitive and specific marker [7]. In this bladder tumor case, negative desmin and S100 staining help distinguish EGIST from leiomyoma and neurogenic tumors; the absence of keratin and smooth muscle actin in pathological sections rules out leiomyoma and leiomyosarcoma, and the negativity for pan-cytokeratin argues against an epithelioid type [15].
Complete surgical resection is the standard treatment for localized GISTs. The goal is R0 surgery, which involves complete tumor excision, including the intact pseudocapsule [1]. Radical cystectomy can perfectly achieve the goal of R0 surgery. GISTs, due to their relatively clear pathogenic mechanisms, can be targeted by imatinib to inhibit the pathological activation of tyrosine kinases caused by Kit/PDGFRA mutations. Imatinib, as a first-line treatment for initially unresectable patients, has ushered in a new era of targeted therapy for stromal tumors [16]. Although complete surgical resection is feasible in most localized GISTs, about 40% of patients will experience metastatic recurrence. Three randomized phase III clinical trials have assessed the role of imatinib adjuvant therapy in preventing disease recurrence and improving Overall Survival (OS). Therefore, this case has indications for adjuvant therapy with imatinib.
The prognosis of EGIST is closely related to tumor size and mitotic activity [17]. According to the 2023 guidelines, these two continuous variables are defined as categorical variables, with the mitotic activity threshold being 5 mitoses per 50 High Power Fields (HPF) and the critical values for tumor size being 2, 5, and 10 cm [1]. In this pathological case, the nuclear mitotic figure is approximately 3/50 HPF, and the maximum tumor diameter is about 7 cm, indicating a high-risk status. This case adopted a robot-assisted laparoscopic radical cystectomy with ileal conduit surgery. EGISTs are considered unresponsive to conventional radiotherapy and chemotherapy, and the patient refused targeted therapy due to economic factors, hence no additional adjuvant therapy was administered postoperatively. The prognosis of GISTs at different sites also affects outcomes, but cases of gastrointestinal stromal tumors originating from the bladder, esophagus, and colorectum are rare, requiring more data to assess risks. In this case, the tumor was large in volume and exhibited full-thickness invasion of the bladder. Imaging studies suggested that the lesion was confined to the anterior wall of the bladder, with suspicious lymphatic metastasis, but no evidence of distant metastasis. However, intraoperatively, we found that the tumor was closely adherent to the abdominal wall muscles and had extensively invaded the extra-bladder fat, though it did not invade the intestines or other organs. We will closely follow up on the patient’s postoperative condition. The efficacy of radical cystectomy for bladder-originating EGIST also requires further support from more clinical cases.
Conclusion
EGIST originating from the bladder are extremely rare. In this report, we describe a case of EGIST with a huge tumor volume and full-thickness bladder infiltration, without lymphatic and distant metastases. For bladder EGIST, surgical treatment is the first choice, and total cystectomy is recommended. Appropriate additional treatments should be carried out after the operation according to the pathological parameters, immunohistochemical parameters and gene testing results.
Declarations
Conflicts of interest: The authors have no conflicts of interest to declare.
Acknowledgments: No funding has been provided for the research.
References
- Serrano C, Martín-Broto J, Asencio-Pascual JM, López-Guerrero JA, Rubió-Casadevall J, Bagué S, et al. 2023 GEIS Guidelines for gastrointestinal stromal tumors. Ther Adv Med Oncol. 2023; 15: 17588359231192388.
- Lasota J, Carlson JA, Miettinen M. Spindle cell tumor of urinary bladder serosa with phenotypic and genotypic features of gastrointestinal stromal tumor. Arch Pathol Lab Med. 2000; 124: 894-7.
- Krokowski M, Jocham D, Choi H, Feller AC, Horny HP. Malignant extragastrointestinal stromal tumor of bladder. J Urol. 2003; 169: 1790-1.
- Mekni A, Chelly I, Azzouz H, Ben Ghorbel I, Bellil S, Haouet S, et al. Extragastrointestinal stromal tumor of the urinary wall bladder: case report and review of the literature. Pathologica. 2008; 100: 173-5.
- García-Segui A, Gascón M. [Gastrointestinal stromal tumour in the urinary bladder]. Actas Urol Esp. 2011; 35: 124-5.
- Shin HS, Cho CH, Kum YS. Extragastrointestinal stromal tumor of the urinary bladder: a case report. Urol J. 2011; 8: 165-7.
- He F, Fang Z, Zhu P, Huang W, Li L. Bladder extragastrointestinal stromal tumor in an adolescent patient: A case-based review. Mol Clin Oncol. 2014; 2: 960-2.
- Hori M, Maki K, Okuno Y, Mikami K, Takeuchi T. A radiation-related second primary gastrointestinal stromal tumor in the bladder. Curr Urol. 2023; 17: 268-70.
- Fujimi A, Nagamachi Y, Yamauchi N, Tamura F, Kimura T, Miyajima N, et al. Gastrointestinal Stromal Tumor in a Patient with Neurofibromatosis Type 1 That Was Successfully Treated with Regorafenib. Intern Med. 2019; 58: 1865-70.
- Lee JN, Kwon SY, Kim J, Yoon GS, Yoo ES, Kim HT. Extra-gastrointestinal stromal tumor on the inner urinary bladder wall. 2016; 14: 43-6.
- Nishida T, Blay JY, Hirota S, Kitagawa Y, Kang YK. The standard diagnosis, treatment, and follow-up of gastrointestinal stromal tumors based on guidelines. Gastric Cancer. 2016; 19: 3-14.
- Lasota J, Miettinen M. Clinical significance of oncogenic KIT and PDGFRA mutations in gastrointestinal stromal tumours. Histopathology. 2008; 53: 245-66.
- Ricci R, Giustiniani MC, Gessi M, Lanza P, Castri F, Biondi A, et al. Telocytes are the physiological counterpart of inflammatory fibroid polyps and PDGFRA-mutant GISTs. J Cell Mol Med. 2018; 22: 4856-62.
- Kondo J, Huh WJ, Franklin JL, Heinrich MC, Rubin BP, Coffey RJ. A smooth muscle-derived, Braf-driven mouse model of Gastrointestinal Stromal Tumor (GIST): evidence for an alternative GIST cell-of-origin. J Pathol. 2020; 252: 441-50.
- Addley S, Alazzam M, Johnson C, Soleymani Majd H. Rectovaginal Extragastrointestinal Stromal Tumour (EGIST): an additional entity to be considered in the differential diagnosis of tumours of the rectovaginal septum. BMJ Case Rep. 2021; 14: e240019.
- Xie F, Luo S, Liu D, Lu X, Wang M, Liu X, et al. Genomic and transcriptomic landscape of human gastrointestinal stromal tumors. Nat Commun. 2024; 15: 9495.
- Miettinen M, Lasota J. Gastrointestinal stromal tumors: pathology and prognosis at different sites. Semin Diagn Pathol. 2006; 23: 70-83.